PUBLIC DISCLOSURE & CLARIFICATION
BioGlyco supplement products (including BioGlyco Flow, BioGlyco Capsules, and BioGlyco Blood) are not affiliated with CD BioGlyco.
CD BioGlyco does not manufacture, distribute, or provide after-sales support for these consumer products. For any product-related questions, please contact the original seller directly.

- Home
- Platforms
- Glyco™ Synthesis Platform
- Custom Glycoprotein Synthesis
- Custom Glycoside Synthesis
- Custom Glycosyl Donor Synthesis
- Custom Thioglycoside Synthesis
- Custom Phosphatidylinositol Synthesis
- Custom Cyclodextrin Synthesis
- Custom Rhamnolipid Synthesis
- Custom Sphingolipid Synthesis
- Custom Building Block Synthesis
- Carbohydrate Manufacturing
- Synthesis Process Development and Optimization
- Custom Carbohydrate Synthesis
- Custom Glycolipid Synthesis
- Custom Glycopeptide Synthesis
- Custom Glycoconjugate Synthesis
- Custom Sugar-Nucleotide Synthesis
- Nucleoside & Nucleotide Synthesis Service
- Custom Oligonucleotide Synthesis Service
- Polynucleotide Synthesis Service
- Nucleoside & Nucleotide Modification Service
- Oligonucleotide Modification Service
- Nucleoside-based Production Service
- Nucleotide-based Production Service
- OPME-based NDP-sugar Synthesis Service
- OPME-based NMP-sugar Synthesis Service
- Glycomics Platform
- Glycoproteomics Platform
- N-Glycoproteomics of PDX Models
- Nanotechnologies for the Detection of Glycopeptides
- Glycoprotein Enrichment
- Glycoprotein Quantification
- Glyco-biomarker Detection Service
- Glyco-biomarker AFP Detection Service
- Glyco-biomarker CA19-9 Detection Service
- Glyco-biomarker CA125 Detection Service
- Glyco-biomarker CA15–3 Detection Service
- Glyco-biomarker CEA Detection Service
- Glyco-biomarker PSA Detection Service
- Glyco-biomarker Immunoglobulin G (IgG) Detection Service
- Glyco-biomarker Haptoglobin (Hp) Detection Service
- Glyco-biomarker α1-Antitrypsin (A1AT) Detection Service
- Glyco-biomarker α1-Acid Glycoprotein (AGP) Detection Service
- Glyco-biomarker Ceruloplasmin (CP) Detection Service
- Glyco-biomarker Fetuin A Detection Service
- Site Occupation
- Glycoprotein Structure Analysis
- Glycosylation Site-specific Antibody-Drug Conjugate (ADC) Development Platform
- Carbohydrate-based Glycomedicine Development Platform
- By Sources of Carbohydrate
- By Types of Carbohydrate
- Monosaccharides-based Glycomedicine Development
- Disaccharides-based Glycomedicine Development
- Trisaccharides-based Glycomedicine Development
- Oligosaccharides-based Glycomedicine Development
- Polysaccharides-based Glycomedicine Development
- Glycosides-based Glycomedicine Development
- Glycopeptide-based Glycomedicine Development
- Glycomimetic-based Glycomedicine Development
- By Applications of-based Glycomedicines
- Carbohydrate-based Anticoagulant Glycomedicine Development
- Carbohydrate-based Cardiovascular Glycomedicine Development
- Carbohydrate-based Antitumor Glycomedicine Development
- Carbohydrate-based Antidiabetic Glycomedicine Development
- Carbohydrate-based Antibacterial Glycomedicine Development
- Carbohydrate-based Antiviral Glycomedicines Development
- Carbohydrate-based Antiparasiti Glycomedicine Development
- By Discovery Strategies
- Glycoengineering-based Glycomedicine Development
- Production of Food Ingredients
- Multi-omics Platform for Cancer Glucose Metabolism (MOPCGM)
- Cancer Cell Glycolytic Analysis
- Cancer Cell TCA Cycle Analysis
- Cancer Cell PPP Analysis
- Gene-level Regulation Analysis of Cancer Glucose Metabolism
- Interplay Between Glucose Metabolism Reprogramming and Tumorigenesis & Progression
- Interplay Between Glucose Metabolism Reprogramming and Proliferative Signaling
- Interplay Between Glucose Metabolism Reprogramming and Growth Suppressor
- Interplay Between Glucose Metabolism Reprogramming and Cancer Cell Death
- Interplay Between Glucose Metabolism Reprogramming and Replicative Immortality
- Interplay Between Glucose Metabolism Reprogramming and Angiogenesis
- Interplay Between Glucose Metabolism Reprogramming and Invasion & Metastasis
- Interplay Between Glucose Metabolism Reprogramming and Immune Escape
- Interplay Between Glucose Metabolism Reprogramming and Genomic Instability
- Interplay Between Glucose Metabolism Reprogramming and Tumor-associated Inflammation
- Glucose Metabolism-Microenvironment Crosstalk Analysis in Cancer
- Cancer Glucose Metabolism In Vivo Analysis
- Applications of Studing Cancer Glucose Metabolism
- Discovery of Cancer Glucose Metabolism Small Molecule Inhibitor
- Discovery of Cancer Glucose Metabolism Therapeutic Monoclonal Antibodies
- Development of Gene Therapy Targeting Cancer Glucose Metabolism
- Discovery of Cancer Glucose Metabolism CAR-T Therapy Targeting
- Discovery of Cancer Glucose Metabolism Combination Therapy Strategy
- Discovery of Cancer Glucose Metabolism Precision Therapeutic Strategies
- Glycoengineering Platform
- Therapeutic Nucleic Acid Development Platform
- Therapeutic Oligonucleotide Synthesis Service
- siRNA Synthesis
- miRNA Synthesis
- ASO Synthesis
- Aptamer Synthesis
- PMO Synthesis
- sgRNA Synthesis
- Circular RNA Synthesis
- PNA Synthesis
- Lipid-based GalNac-RNA Delivery Service
- CpG Oligonucleotide Synthesis
- Click-based GalNac-RNA Delivery Service
- AntimiR Synthesis
- GalNAc-siRNA Delivery Service
- tRNA Synthesis
- GalNAc-ASO Delivery Service
- saRNA Synthesis
- Reporter Gene mRNA Synthesis
- Gene Editing mRNA Synthesis
- Gene Replacement mRNA Synthesis
- Cre Recombinase mRNA Synthesis
- hEPO mRNA Synthesis
- OVA mRNA Synthesis
- Therapeutic Oligonucleotide Modification Service
- Therapeutic Oligonucleotide Delivery Development Service
- Targeted Ligand and Linker Synthesis
- Monoantennary GalNac-RNA Delivery
- Biantennary GalNac-RNA Delivery
- Triantennary GalNac-RNA Delivery
- Tetra-antennary GalNac-RNA Delivery
- Solution Phase-based GalNac-RNA Delivery
- Solid Phase-based GalNac-RNA Delivery
- mRNA Poly(A) Tail Length Analysis
- GalNAc-siRNA Delivery
- GalNAc-ASO Delivery
- GalNAc-miRNA Delivery
- GalNAc-Aptamer Delivery
- GalNAc-AntimiR Delivery
- GalNAc-mRNA Delivery
- GalNAc-PNA Delivery
- Peptide-Therapeutic Oligonucleotide Delivery
- LNP-Therapeutic Oligonucleotide Delivery
- PEG-Therapeutic Oligonucleotide Delivery
- Biological Evaluation Service for Therapeutic Oligonucleotide
- mRNA-based Vaccine Development Service
- mRNA Sequence Design&Optimization Service
- mRNA Integrity Analysis ServicemRNA Integrity Analysis Service
- mRNA Biodistribution Analysis Service
- DNA Template Preparation Service
- mRNA Purity Analysis Service
- T Cell Cytokine Secretion Analysis Service
- mRNA Modification Service
- mRNA Length Analysis Service
- mRNA Purification Service
- Neutralizing Antibody Assay Service
- mRNA-LNP Formulation and Encapsulation Service
- mRNA Structural Characterization
- mRNA Structural Characterization Service
- mRNA Bioanalysis Service
- mRNA Residual DNA Template Analysis Service
- mRNA Residual Double-Stranded RNA (dsRNA) Analysis Service
- mRNA Size Analysis Service
- mRNA Polydispersity Analysis Service
- mRNA Zeta Potential Analysis Service
- mRNA Lipid Composition Analysis Service
- Oligonucleotide Drug Process and Formulation Development Service
- RNA Drug Process and Formulation Development
- Therapeutic Oligonucleotide Synthesis Service
- Glycobiology Microarray Platform
- Glyco™ Vaccine Development Platform
- Carbohydrate-based Vaccine Development
- Polysaccharide Vaccine Development
- Tumor-Associated Carbohydrate Vaccine Development
- Glycoconjugate Vaccine Development
- Natural Carbohydrate-based Vaccine Development
- Semi-synthetic Carbohydrate-based Vaccine Development
- Fully Synthetic Carbohydrate-based Vaccine Development
- Carbohydrate-based Antibacterial Vaccine Development
- Carbohydrate-based Antifungal Vaccine Development
- Carbohydrate-based Antiparasitic Vaccine Development
- Carbohydrate-based Antiviral Vaccine Development
- Carbohydrate-based Anticancer Vaccine Development
- Carbohydrate-based Antihelmintic Vaccine Development
- Monovalent Carbohydrate-based Vaccine Development
- Polyvalent Carbohydrate-based Vaccine Development
- Carrier Protein Design Service
- Carbohydrate Conjugation Service for Vaccine Development
- Carbohydrate-based Adjuvant Development
- Lipopolysaccharide-based Adjuvant Development
- Bacterial Outer Membrane Vesicles (OMVs)-based Adjuvant Development
- Trehalose Glycolipid-based Adjuvant Development
- Galactosylceramide-based Adjuvant Development
- Peptidoglycan-based Adjuvant Development
- Chitin/Chitosan-based Adjuvant Development
- Inulin-based Adjuvant Development
- Mannans-based Adjuvant Development
- Alginate-based Adjuvant Development
- Saponin-based Adjuvant Development
- α-Glucan-based Adjuvant Development
- Lentinan-based Adjuvant Development
- β-Glucans-based Adjuvant Development
- Deltin-based Adjuvant Development
- Muramyldipeptide-based Adjuvant Development
- Cord Factor-based Adjuvant Development
- Zwitterionic Polysaccharide-based Adjuvant Development
- Novel Plant Polysaccharides-based Adjuvant Development
- Glycoprotein-based Vaccine Development
- Glycopeptide-based Vaccine Development
- Carbohydrate-based Vaccine Development
- Glycogenomics Platform
- Glycogene Editing Service
- Glycogene Delivery Service
- Glycogene Expression Profiling
- Cancer Glycogene Discovery Service
- Glycogene Discovery Service in Cervical Cancer
- Glycogene Discovery Service in Leukemia
- Glycogene Discovery Service in Bladder Cancer
- Glycogene Discovery Service in Colorectal Adenocarcinoma
- Glycogene Discovery Service in Liver Cancer
- Glycogene Discovery Service in Lymphoma
- Glycogene Discovery Service in Breast Cancer
- Glycogene Discovery Service in Prostate Cancer
- Glycogene Discovery Service in Pancreatic Cancer
- Glycogene Discovery Service in Lung Cancer
- Glycogene Discovery Service in Thyroid Cancer
- Glycogene Discovery Service in Ovarian Cancer
- Glycan Display Platform
- Traditional Glycan Display Array
- Natural Oligosaccharide Library Construction Service
- Chemical Synthesis-based Oligosaccharide Library Construction Service
- Enzymatic Synthesis-based Oligosaccharide Library Construction Service
- Modular Synthesis-based Oligosaccharide Library Construction Service
- Oligosaccharide Library Immobilization Service
- Cell-based Glycan Display Array
- Neoglycolipid (NGL) Display Array
- Liquid Glycan Display Array (LiGA)
- Glycophage Display
- N-linked Glycoprotein-based Glycophage Display System Construction Service
- O-linked Glycoprotein-based Glycophage Display System Construction Service
- Glycophage Display-based Glycosylase Genetic Analysis Service
- Glycophag Display-based Glycoarray Service
- Glycophage Display-based Antibody Development Service
- Glycophage Display-based Epitope Mapping Service
- Glycophage Display-based Biomarker Development Service
- De Novo Glycan Display
- Cell-Surface Glycan Editing
- Examining the Effects of Altering Blood Group Antigens on Erythrocyte Cell Surfaces
- Preventing Cellular Rejection During Transplantation
- Modulating Chemical Composition and Physical Parameters of Glycocalyx
- Tailoring Cell Membranes with Biologically Active Glycans
- Targeting Glycosaminoglycan-mediated Growth Factor Signaling to Influence Stem Cell Specification
- Long-term De Novo Glycan Display for Directing Stem Cell Fate
- Traditional Glycan Display Array
- GlycoNano™ Platform
- Glyconanoparticle Development Service
- Techniques for Glyconanoparticle Development
- Carbohydrate-based Nanoparticle Production
- Gold Glyconanoparticle Production
- Silver Glyconanoparticle
- Magnetic Glyconanoparticle
- Quantum Dot (QD) Glyconanoparticle
- Glyconanoparticle Characterization
- PEG Glyconanoparticle
- Carbon-based Glyconanoparticle
- Fluorescent Glyconanoparticle
- Silica Glyconanoparticle
- Liposome Glyconanoparticle
- Glycol Nanohydrogel Development
- Glycol Nanorod
- Glycol Nanotube
- Glycol Nanocrystal/Nanosheet/Nanosphere/Nanofiber
- Glyconanoparticle Preclinical Study
- Glyconanoparticle Formulation
- Glyconanotechnology-based Biosensor Development
- GlycoNano™ Bioink Production for 3D Printing
- Glyconanoparticle Development Service
- Glyco™ Synthesis Platform
- Services
- Fatty Acid Analysis
- Omega-3 Fatty Acid Analysis
- Omega-6 Fatty Acid Analysis
- Esterified Fatty Acid Analysis
- Saturated Fatty Acid Analysis
- Straight-chain Fatty Acid Analysis
- Volatile Fatty Acid Analysis
- Cis/Trans Fatty Acid Analysis
- Whole-Cell Fatty Acid Analysis
- Free Fatty Acid Analysis
- Total Fatty Acid Analysis
- Long-chain Fatty Acid Analysis
- Short-chain Fatty Acid Analysis
- Glycosylation Inhibitor Development
- Metabolic Interconversion Inhibitor Development
- N-Glycosylation Inhibitor Development
- O-Glycosylation Inhibitor Development
- O-GalNAc Inhibitor Development
- Core 1 Inhibitor Development
- Core 2 Inhibitor Development
- Core 3 Inhibitor Development
- Core 4 Inhibitor Development
- Sialyl-Tn Antigen Inhibitor Development
- Sialyl-T Antigen Inhibitor Development
- SLex Antigen Inhibitor Development
- Blocking O-Glycan-Protein Interaction Inhibitor Development
- Capping Modification Inhibitor Development
- Postsynthetic Glycan Modification Inhibitor Development
- GAG Inhibitor Development
- GSL Inhibitor Development
- GPI Anchor Inhibitor Development
- O-GlcNAc Inhibitor Development
- Lipidomics Analysis Services
- Carbohydrate Separation, Purification and Analysis
- Carbohydrate Metabolism Analysis
- Monosaccharide Analysis Service
- Mannitol Analysis Service
- Mannose Analysis Service
- Fructose Analysis Service
- Xylose Analysis Service
- Glucose Analysis Service
- Sorbitol Analysis Service
- Galactose Analysis Service
- Fucose Analysis Service
- Arabinose Analysis Service
- Ribose Analysis Service
- Rhamnose Analysis Service
- Galacturonic Acid Analysis Service
- Glucuronic Acid Analysis Service
- Galactosamine Analysis Service
- Mannuronic Acid Analysis Service
- Guluronic Acid Analysis Service
- Mannosamine Analysis Service
- Glucosamine Analysis Service
- Muramic Acid Analysis Service
- N-Acetylglucosamine Analysis Service
- Tagatose Analysis Service
- Talose Analysis Service
- Psicose Analysis Service
- Sorbose Analysis Service
- Disaccharide Analysis Service
- Maltose Analysis Service
- Isomaltose Analysis Service
- Lactose Analysis Service
- Sucrose Analysis Service
- Trehalose Analysis Service
- Melibiose Analysis Service
- Turanose Analysis Service
- Lactulose Analysis Service
- Maltitol Analysis Service
- Lactitol Analysis Service
- Epilactose Analysis Service
- Cellobiose Analysis Service
- Allolactose Analysis Service
- Maltotriose Analysis Service
- Isomaltulose Analysis Service
- Oligosaccharide Analysis Service
- Raffinose Analysis Service
- Stachyose Analysis Service
- Mannan Oligosaccharides Analysis Service
- Xylooligosaccharide Analysis Service
- Fructooligosaccharide Analysis Service
- Galactooligosaccharide Analysis Service
- Chitooligosaccharide Analysis Service
- Arabinoxylooligosaccharide Analysis Service
- Pectic Oligosaccharide Analysis Service
- Maltooligosaccharide Analysis Service
- Isomaltooligosaccharide Analysis Service
- Polysaccharide Analysis Service
- Sea Cucumber Polysaccharide Analysis Service
- Lycium barbarum Polysaccharide Analysis Service
- Polygonatum Polysaccharide Analysis Service
- Glycogen Analysis Service
- Glucomannan Analysis Service
- Chitin/Chitosan Analysis Service
- Glucan Analysis Service
- Galactan Analysis Service
- Xylan Analysis Service
- Mannan Analysis Service
- Arabinan Analysis Service
- Rhamnan Analysis Service
- Fucoidan Analysis Service
- Galactomannan Analysis Service
- Glycosaminoglycan Analysis Service
- Monosaccharide Analysis Service
- Custom Glycosylation Service
- Sialic Acid Analysis Service
- Strategies for Sialic Acid Analysis
- Aberrant Sialylation in Cancers
- Sialic Acid Analysis in Pancreatic Cancer
- Sialic Acid Analysis in Laryngeal Cancer
- Sialic Acid Analysis in Oral Cancer
- Sialic Acid Analysis in Cholangiocarcinoma
- Sialic Acid Analysis in Cervical Cancer
- Sialic Acid Analysis in Leukemia
- Sialic Acid Analysis in Colorectal Cancer
- Sialic Acid Analysis in Lung Cancer
- Sialic Acid Analysis in Breast Cancer
- Sialic Acid Analysis in Endometrial Cancer
- Sialic Acid Analysis in Stomach Cancer
- Sialic Acid Analysis in Thyroid Cancer
- Sialic Acid Analysis in Malignant Melanoma
- Sialic Acid Analysis in Prostate Cancer
- Sialic Acid Analysis in Ovarian Cancer
- Custom Sialyl Tumor Antigen Production Service
- High-Throughput Glycan Screening Service
- HTS of Important Glycans
- HTS of Glycan Derivatives
- HTS for Glycan-Binding Proteins
- HTS for Compounds That Alter Muscle Cell Glycosylation
- HT Glycan Screening for Antibodies
- HT Glycan Screening for Glycoengineered Cell Line
- HTS for Human Galactokinase Inhibitors
- HTS for Inhibitors of Sialyl- and Fucosyltransferases
- HT Automated Micro-Permethylation for Glycan Structure Analysis
- Strategies for HT Glycan Screening
- HT Glycan Screening Using Microchip-Based Assay
- HT Glycan Screening Using Miniature Pig Kidney N-Glycan-Immobilized Beads
- HT Glycan Screening Using An Automated LC-MS-Based Solution
- HT Glycan Screening Using Mass Spectrometry
- HT Glycan Screening Using UHPLC-FLD
- HT Glycan Screening Using 19F-Labeled Probes
- HT Glycan Screening Using Hydrophilic Interaction Chromatography
- HT Glycan Screening Using MALDI-TOF-MS
- HT Glycan Screening Using CE-MS
- HT Glycan Screening Using HPLC-Based Analysis
- HT Glycan Screening Using ESI-MSn
- HT Glycan Screening Using a DNA Sequencer
- HT Glycan Screening Using Glycan Microarrays
- HT Glycan Screening Using a 96-Well Plate Platform
- Characterization of Glycosylation in Drugs
- Glycobiology of Human Milk
- Anti-glycan Antibody Development
- Lectin Production Service
- Pharmaceutical and Biological Analysis Service
- Pharmaceutical Analysis Service
- Oral Rehydration Salt Analysis Service
- Pentoxifylline and Glucose Injection Analysis Service
- Xylitol Analysis Service
- Ulinastatin Analysis Service
- Deslanoside Analysis Service
- Glycerol Fructose and Sodium Chloride Injection Analysis Service
- Josamycin Propionate Analysis Service
- Dextran Analysis Service
- Iron Dextran Analysis Service
- Capecitabine Analysis Service
- Metildigoxin Analysis Service
- Metronidazole and Glucose Injection Analysis Service
- Gadobenate Dimeglumine Injection Analysis Service
- Voglibose Analysis Service
- Inosine and Glucose Injection Analysis Service
- Heparin Calcium Analysis Service
- Heparin Sodium Analysis Service
- Acarbose Analysis Service
- Amikacin Analysis Service
- Aciclovir and Glucose Injection Analysis Service
- Streptomycin Injection Analysis Service
- Erythromycin Lactobionate Analysis Service
- Saccharated Pepsin Analysis Service
- Compound Sodium Lactate and Glucose Injection Analysis Service
- Tylosin Analysis Service
- Compound Calcium Gluconate Oral Solution Analysis Service
- Cytarabine Hydrochloride for Injection Analysis Service
- Oxystarch Analysis Service
- Glucosamine Indometacin Enteric Capsule Analysis Service
- Glucose and Sodium Chloride Injection Analysis Service
- Ferrous GIuconate Analysis Service
- Calcium Gluconate Analysis Service
- Calcium Gluconate and Sodium Chloride Injection Analysis Service
- Zinc Gluconate Analysis Service
- Sodium Stibogluconate Analysis Service
- Chlorhexidine Gluconate Analysis Service
- Sodium Stibogluconate Injection Analysis Service
- Kanamycin Sulfate Analysis Service
- Gentamicin Sulfate Analysis Service
- Isepamicin Sulfate Analysis Service
- Amikacin Sulfate Analysis Service
- Chondroitin Sulfate Sodium Analysis Service
- Etimicin Sulfate Analysis Service
- Ribostamycin Sulfate Analysis Service
- Streptomycin Sulfate Analysis Service
- Neomycin Sulfate Analysis Service
- Sucralfate Analysis Service
- Potassium Chloride and Glucose Injection Analysis Service
- Anticoagulant Citrate Dextrose Analysis Service
- Aurothioglucose Analysis Service
- Calcium Gluceptate Analysis Service
- Calcium Lactobionate Analysis
- Calcium Levulinate Analysis
- Oxidized Cellulose Analysis
- Oxidized Regenerated Cellulose Analysis
- Cellulose Sodium Phosphate Analysis
- Chlorpromazine Hydrochloride Syrup Analysis
- Clofibrate Capsule Analysis
- Copper Gluconate Analysis
- Cytarabine Analysis
- Dextrose Analysis
- Diatrizoate Meglumine Analysis
- Digitoxin Analysis
- Digoxin Analysis
- Dihydrostreptomycin Sulfate Analysis
- Dirithromycin Analysis
- Enoxaparin Sodium Analysis
- Erythromycin Analysis
- Erythromycin Estolate Analysis
- Erythromycin Ethylsuccinate Analysis
- Sterile Erythromycin Gluceptate Analysis
- Ferrous Sulfate Syrup Analysis
- Fludeoxyglucose F 18 Injection Analysis
- Beta Glucan Analysis
- Gentamicin and Prednisolone Acetate Ophthalmic Ointment Analysis
- Gluconolactone Analysis
- Heparin Lock Flush Solution Analysis
- Anticoagulant Heparin Solution Analysis
- Hyaluronidase Injection Analysis
- Iodipamide Meglumine Injection Analysis
- Iron Sorbitex Injection Analysis
- Iron Sucrose Injection Analysis
- Magnesium Gluconate Analysis
- Manganese Gluconate Analysis
- Meglumine Analysis
- Natamycin Analysis
- Quinidine Gluconate Analysis
- Sisomicin Sulfate Analysis
- Sodium Gluconate Analysis
- Biological Evaluation
- Typhoid Vaccine Evaluation
- RNA-Cell Binding Interaction Analysis Services
- Group A Meningococcal Polysaccharide Vaccine Evaluation
- Off-Target Activity Assessment Service
- 23-Valent Pneumococcal Polysaccharide Vaccine Evaluation
- Conjugate Potency Assay Service
- H. influenzae Type b Conjugate Vaccine Evaluation
- ADME Profiling Service
- Poliomyelitis Vaccine in Dragee Candy Evaluation
- Human Hepatitis B Immunoglobulin Evaluation
- Human Rabies Immunoglobulin Evaluation
- Human Tetanus Immunoglobulin Evaluation
- Human Immunoglobulin Evaluation
- Anti-human T Lymphocyte Rabbit Immunoglobulin Evaluation
- Human Fibrinogen Evaluation
- Human Thrombin Evaluation
- Conbercept Ophthalmic Injection Evaluation
- BCG-PPD Evaluation
- BR-PPD Evaluation
- Pharmaceutical Excipient Analysis
- Ethylcellulose Analysis Service
- Sucralose Analysis
- Wheat Starch Analysis
- Potato Starch Analysis
- Tapioca Starch Analysis
- Maize Starch Analysis
- Compressible Sugar Analysis
- Soluble Starch Analysis
- Methylcellulose Analysis
- Guar Gum Analysis
- Tragacanth Analysis
- Croscarmellose Sodium Analysis
- Cellacefate Analysis
- Maltodextrin Analysis
- Low-substituted Hydroxypropyl Cellulose Analysis
- Alpha Cyclodextrin Analysis
- Beta Cyclodextrin Analysis
- Gamma Cyclodextrin Analysis
- Arabino Galactan Analysis
- Acacia Analysis
- Pectin Analysis
- Powdered Cellulose Analysis
- Alginic Acid Analysis
- Sodium Alginate Analysis
- Pregelatinized Hydroxypropyl Starch Analysis
- Pregelatinized Starch Analysis
- Xanthan Gum Analysis
- Steviol Glycoside Analysis
- Hydroxyethyl Cellulose Analysis
- Hypromellose Analysis
- Hydroxypropyl Cellulose Analysis
- Hydroxypropyl Betadex Analysis
- Vacant HydroxypropyI Starch Capsule Analysis
- Dextrate Analysis
- Agar Analysis
- Calcium Saccharate Analysis
- Caramel Analysis
- Pullulan Analysis
- Microcrystalline Cellulose Analysis
- Carboxymethylcellulose Calcium Analysis
- Carboxymethylcellulose Sodium Analysis
- Sodium Starch Glycolate Analysis
- Polydextrose Analysis
- Sucrose Octaacetate Analysis
- Sugar Sphere Analysis
- Sucrose Stearate Analysis
- Pea Starch Analysis
- Cellulose Acetate Analysis
- Hypromellose Acetate Succinate Analysis
- Dextrin Analysis
- Sodium Starch Phosphate Analysis
- Gellan Analysis
- Hyaluronan Analysis
- Pharmaceutical Analysis Service
- Carbohydrate-based Biodegradable Material Development
- Monosaccharide-based Biodegradable Material Development
- Disaccharide-based Biodegradable Material Development
- Oligosaccharide-based Biodegradable Material Development
- Polysaccharide-based Biodegradable Material Development
- Cellulose-based Biodegradable Material Development
- Starch-based Biodegradable Material Development
- Chitosan-based Biodegradable Material Development
- Dextran-based Biodegradable Material Development
- Hyaluronic Acid-based Biodegradable Material Development
- Other Polysaccharide-based Biodegradable Material Development
- Modification of Biodegradable Polysaccharide
- Characterization of Polysaccharide-based Biodegradable Material
- Applications of Polysaccharide-based Biodegradable Material
- Glycobiology Disease Model Development Service
- Techniques for In Vivo Glycobiology Disease Model Development
- Gene Editing-based In Vivo Glycobiology Disease Model Development Service
- Transgenic-based In Vivo Glycobiology Disease Model Development Service
- Embryo Implantation and Cultivation-based In Vivo Glycobiology Disease Model Development Service
- Selective Breeding-based In Vivo Glycobiology Disease Model Development Service
- Gene Expression-based In Vivo Glycobiology Disease Model Development Service
- Glycobiology Disease Model Construction Service
- Custom In Vivo Glycobiology Disease Model Service
- In Vivo Glycobiology Disease Model Screening Service
- In Vitro Glycobiology Disease Model Screening Service
- HeLa Cells-based In Vivo Screening Service
- 3T3-L1 Cells-based In Vitro Screening Service
- Muscle Cells-based In Vitro Screening Service
- Hepatocyte-based In Vitro Screening Service
- Insulin Target Cells-based In Vitro Screening Service
- Intestinal Cells-based In Vitro Screening Service
- THP-1 Monocyte-based In Vitro Screening Service
- Vero Cells-based In Vitro Screening Service
- Techniques for In Vivo Glycobiology Disease Model Development
- Fatty Acid Analysis
- Products
- Hot Glycans
- Featured Products
- Carbohydrates
- Nucleosides and Nucleotides
- Enzymes
- Lectin Products
- Carbohydrate Antigen
- Glycoconjugates
- Assay Kits
- Glycosylation Related Products
- Natural Products
- Inhibitors
- Protein Tyrosine Kinases
- TGF-β-Smad
- Cell Cycle
- Neuronal Signaling
- Proteases
- Cell Metabolism
- Endocrinology-Hormones
- GPCR-G Protein
- Adrenergic Receptor
- Histamine Receptor
- mGluR
- 5-HT Receptor
- Adenosine Receptor
- Dopamine Receptor
- Neurokinin Receptor
- Calcium-sensing Receptor
- S1P Receptor
- Platelet-activating Factor (PAF) Receptor
- LPA Receptor
- Smoothened Receptor
- Opioid Receptor
- Melatonin Receptor
- CGRP Receptor
- Endothelin Receptor
- Prostanoid Receptor
- Cannabinoid Receptor
- Glucagon Receptor
- G Protein
- Guanylate Cyclase
- Orexin Receptor
- Imidazoline Receptor
- Neurotensin Receptor
- Peptide Receptor
- CCK
- NPY Receptor
- MCHR1 (GPR24)
- Sigma Receptor
- GHSR
- GPCR19
- Formyl Peptide Receptor (FPR)
- Angiotensin Receptors
- GLP-1 receptors
- Rev-Erb Receptors
- CRFR
- Vasopressin Receptor
- Protease-Activated Receptors (PAR)
- Oxytocin Receptor
- Bradykinin Receptor
- Adenylyl Cyclase
- Urotensin Receptor
- Somatostatin Receptors
- Bombesin Receptor
- cAMP
- CXCR
- Leukotriene Receptor
- Membrane Transporters-Ion Channels
- AMPA Receptor
- Sodium Channel
- Calcium Channel
- SGLT
- ATPase
- Proton Pump
- Potassium Channel
- P-gp
- CFTR
- Chloride Channels
- TRPV
- Monocarboxylate Transporters (MCT)
- HCN Channel
- NCX
- TRP Channel
- Na+-H+ Exchanger
- OCT
- Glutamate (EAAT) Transporter
- Serotonin Transporter
- BCRP
- VDAC
- Aquaporin
- Piezo Channel
- Monoamine Transporter
- CRM1
- Transferases
- Stem Cells-Wnt
- Autophagy Signaling
- Metabolism
- MAO
- PPAR
- FXR
- Cytochrome P450
- PDE
- HSP
- Hydroxylase
- Factor Xa
- DHFR
- Dehydrogenases
- Procollagen C-proteinase
- Carbonic Anhydrase
- LXR
- FAAH
- 15-PGDH
- CETP
- Ferroptosis
- HMG-CoA Reductase
- Isocitrate Dehydrogenase (IDH)
- IDO
- Retinoid Receptor
- Lipase
- Phospholipase
- Lipoxygenase
- AhR
- NAMPT
- GLUT
- Antimetabolite
- Hexokinase
- Epoxide Hydrolase
- Stearoyl-CoA Desaturase (SCD)
- Lipid Metabolism
- Neuronal Metabolism
- SREBP
- LDL
- Foxo1
- DGAT
- Farnesyl Transferase
- Thioredoxin
- Vitamin
- Decarboxylase
- ACLY
- Fatty Acid Synthase (FASN)
- Glucokinase
- Lactate Dehydrogenase A (LDHA)
- Oxidative Phosphorylation
- PFKFB3
- Ribonucleotide Reductase
- Acetyl-CoA Carboxylase
- Glucosidase
- PAI-1
- Immunology & Inflammation
- PI3K/Akt/mTOR
- Angiogenesis
- Apoptosis
- DNA Damage
- Epigenetics
- JAK/Stat
- MAPK
- Microbiology
- NF-kB/IkB
- Stem Cells/Wnt
- DNA-RNA Synthesis
- Cytoskeletal Signaling
- Ubiquitin
- Other inhibitors
- PROTAC
- Bioactive Compounds
- Glycobiology Arrays
- Glycolipids
- Nanomaterials
- Resources
- Contact Us

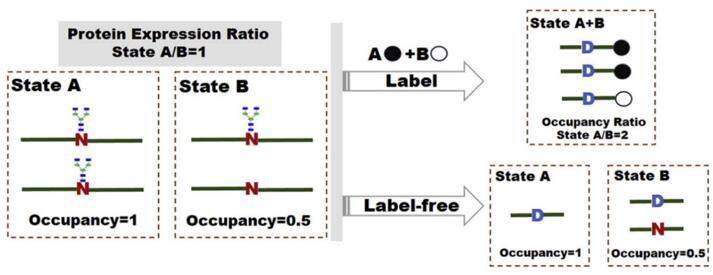 Fig.1 MS-based labeling and label-free technologies for quantification of N-glycosylation site occupancy. (Zhang, 2017)
Fig.1 MS-based labeling and label-free technologies for quantification of N-glycosylation site occupancy. (Zhang, 2017) 




